Обновлено: 09.03.2023
Work in groups. Collect information using the Internet about what we can do to solve the problem of acid rain and present it to the class. Draw pictures to show some of the things we can do.
Решение
Перевод задания
Работа в группах. С помощью Интернета соберите информацию о том, что мы можем сделать, чтобы решить проблему кислотного дождя, и представьте ее классу. Нарисуйте картинки, чтобы показать, на что мы способны.
ОТВЕТ
A great way to reduce acid rain is to produce energy without using fossil fuels. Instead, people can use renewable energy sources, such as solar and wind power. Renewable energy sources help reduce acid rain because they produce much less pollution.
Since energy production creates large amounts of the pollutants that cause acid rain, one important step you can take is to conserve energy. You can do this in a number of ways:
1 )Turn off lights, computers, televisions, video games, and other electrical equipment when you’re not using them.
2 ) Encourage your parents to buy equipment that uses less electricity, including lights, air conditioners, heaters, refrigerators, and washing machines. Such equipment might have the Energy Star label.
3 ) Try to limit the use of air conditioning.
4 ) Ask your parents to adjust the thermostat (the device used to control the temperature in your home) when you go on vacation.
Minimize the Kilometers
Driving cars and trucks also produces large amounts of nitrogen oxides, which cause acid rain. To help cut down on air pollution from cars, you can carpool or take public transportation, such as buses and trains. Also, ask your parents to walk or bike with you to a nearby store or friend’s house instead of driving.
So, purchase energy−saving appliances; carpool, use public transportation, walk, or bike whenever possible; keep your thermostat low in the winter and high in the summer; insulate your house; and turn off lights, computers, and appliances when you’re not using them.
Перевод ответа
Отличный способ уменьшить кислотные дожди − это производить энергию без использования ископаемого топлива. Вместо этого люди могут использовать возобновляемые источники энергии, такие как солнечная и ветровая энергия. Возобновляемые источники энергии помогают уменьшить кислотные дожди, поскольку они производят гораздо меньше загрязнения.
Поскольку при производстве энергии образуется большое количество загрязняющих веществ, вызывающих кислотные дожди, одним из важных шагов, которые вы можете предпринять, является экономия энергии. Вы можете сделать это несколькими способами:
1 )Выключайте свет, компьютеры, телевизоры, видеоигры и другое электрическое оборудование, когда вы им не пользуетесь.
2 ) Поощряйте родителей покупать оборудование, которое потребляет меньше электроэнергии, в том числе свет, кондиционеры, обогреватели, холодильники и стиральные машины. Такое оборудование может иметь маркировку Energy Star.
3 ) Постарайтесь ограничить использование кондиционера.
4 ) Попросите родителей настроить термостат (устройство, которое используется для контроля температуры в вашем доме), когда вы уезжаете в отпуск.
Сведите к минимуму километры пробега
Вождение легковых и грузовых автомобилей также производит большое количество оксидов азота, которые вызывают кислотные дожди. Чтобы сократить загрязнение воздуха от автомобилей, вы можете пользоваться автопарком или общественным транспортом, например автобусами и поездами. Кроме того, попросите родителей пойти с вами пешком или на велосипеде до ближайшего магазина или дома к другу, вместо того, чтобы вести машину.
Итак, приобретите энергосберегающую технику; попутешествуйте, пользуйтесь общественным транспортом, пешком или на велосипеде, когда это возможно; держите термостат низким зимой и высоким летом; утеплить свой дом; и выключайте свет, компьютеры и бытовую технику, когда вы ими не пользуетесь.
Тема по английскому языку: Экологические проблемы
Актуальные проблемы
Э кологические проблемы причиняют огромный вред окружающей среде. Среди самых актуальных – озоновый слой, кислотные дожди, глобальное потепление, токсическое загрязнение атмосферы, исчезновение лесов, загрязнение подземных вод химикатами, разрушение почвы в некоторых областях и угроза представителям флоры и фауны.
Виды загрязнения
Земля является домом для миллиона живых существ, которые составляют сложный мир природы. В наши дни люди пытаются изменить их среду обитания, чтобы удовлетворить свои потребности – создавать сельскохозяйственные угодья или строить города. Они загрязняют и разрушают среду обитания дикой природы, копая землю для угледобычи или строя дороги. Известно, что четверть всех растений находится под угрозой исчезновения. Существуют различные виды загрязнения: водное, воздушное, загрязнение земли и радиоактивное.
Кислотный дождь
Кислотный дождь выпадает, когда ядовитые газы от атомных станций и автомобильных выхлопов смешиваются с кислородом и влагой в воздухе. Эти газы становятся частью водного цикла, и могут долгое время разноситься ветром, пока не выпадут кислотным дождем, который убивает жизнь в озерах, реках и лесах и разрушает жизнь растений.
Глобальное потепление
Мировые температуры повышаются каждый год. Это так называемое глобальное потепление вызвано скоплением газов и испарением воды в атмосфере. Эти газы образуют слой, который отражает тепло назад на землю. Так как планета нагревается, полярные льды начнут таять. Это может вызвать повышение уровня моря, и многие среды обитания исчезнут под водой.
Заключение
Экологические проблемы не имеют границ. Однако, катастроф окружающей среды можно избежать, если люди будут расширять свои экологические знания, и каждый будет понимать, что красота природы очень хрупкая. Правительства должны принимать серьезные меры по борьбе с загрязнением окружающей среды.
Ecological problems
Problems
Ecological problems are causing great damage to our environment. Among the most urgent ones are the ozone layer, acid rains, global warming, toxic pollution of atmosphere, disappearance of forests, contamination of underground waters by chemical elements, destruction of soil in some areas, and threat to some flora and fauna representatives.
Types of pollution
The Earth is a home to millions of different kinds of living things, which make up the complex world of nature. Nowadays people try to change their habitats to suit their own needs – to create farmlands or build cities. They create pollution and destroy wildlife habitats by digging the ground up for mining, or by building roads through them. A quarter of all the plants in the world are known to be in a danger or threatened with extinction. There are different types of pollution: water pollution, air pollution, ground pollution and nuclear pollution.
Acid rain
Acid rain falls when poisonous gases from power stations and vehicle exhausts mix with oxygen and moisture in the air. These gases become part of the water cycle and may be carried a long way by the wind before they fall as acid rain, which kills wildlife in lakes, rivers, and forests, and damages the surrounding plant life.
Global warming
World temperatures are currently rising every year. This so called global warming is caused by the building of gases and water vapour in the atmosphere. These gases form a layer that reflects the heat back to Earth. As the planet warms up, the polar ice caps will start to melt. This could cause sea levels to rise and many habitats will disappear under water.
Conclusion
Ecological problems have no borders. However, environment disasters can be avoided if people broaden ecological education and every person understands that the beauty of nature is extremely fragile. Governments must take serious actions against pollution.
2. a) Look at the pictures again and the headings in the text on p. 77. Can you explain the problem? Read and check. — Посмотрите еще раз на картинки и заголовки текста на странице 77. Вы можете объяснить проблему? Прочитайте и проверьте.
Factories and power stations burn coal, cars burn petrol and emit toxic fumes. This causes air pollution and acid rain. Acid rain falls on the earth, seas, rivers, etc, causing water and soil pollution. A lot of fish and plant species are dying.
Заводы и электростанции сжигают уголь, машины сжигают бензин и выбрасывают токсичные вещества. Это влечет за собой загрязнение воздуха и кислотный дождь. Кислотный дождь падает на землю, моря, реки и т.д., становясь причиной загрязнения воды и почвы. Целые виды рыб и растений вымирают.
b) Read carefully and fill in the blanks with the best word A, B, or C. Listen and check. Then, explain the words in bold. — Прочитайте внимательно и заполните пустые места наиболее подходящим словом вариантов A, B, или С. Послушайте и проверьте. Затем, объясните слова, выделенные жирным шрифтом.
Acid Rain
We all need to work together to stop this problem before it’s too late.
Кислотный дождь
Нам всем нужно работать вместе, чтобы остановить эту проблему, пока не стало слишком поздно.
Using notes to give a talk — Используем тезисы для выступления
When you use notes to give a talk, look at your audience and use your notes to remind you of what you want to say. This will make your talk sound more interesting and natural. — Когда вы используете тезисы для выступления, смотрите на свою аудиторию и используйте тезисы, чтобы вспомнить, что вы хотите сказать. Это сделает вашу речь более интересной и естественной.
Примечание: Авторы хотели сказать, что при выступлении не следует читать текст речи по бумажке. Лучше сделать ключевые тезисы, которые позволят вам вспомнить развернутое содержание темы.
3. Make notes under the headings and give a talk about acid rain. — Составьте тезисы по заголовкам и расскажите о кислотном дожде
В статье перечисляются самые большие экологические проблемы, с которыми сталкивается наша планета сегодня, и краатко поясняется, почему они имеют большое значение для нашей жизни.
1. Climate Change — Изменение климата
Изменение климата — главная экологическая проблема, с которой сегодня сталкивается планета.
Статистика утверждает, что с 1880 года глобальные температуры выросли на 1,7 градуса по Фаренгейту, что напрямую связано с сокращением арктического льда на 13,3% за десятилетие.
Последствия изменения климата широко распространены, поскольку это вызовет проблемы с обезлесением, водоснабжением, океанами и экосистемами.
Изменение климата и многие факторы, способствующие выбросам, могут привести к катастрофическим проблемам в будущем.
Необходимо приложить больше усилий, чтобы устранить основные экологические проблемы, которые затрагивают нас сегодня. Если этого не произойдет, существует вероятность, что большие участки планеты станут необитаемыми в будущем.
Хорошей новостью является то, что многие из этих проблем можно контролировать. Внося коррективы, человечество может оказывать прямое и положительное влияние на окружающую среду.
2. Polar Ice Caps — Полярные льды
Вопрос о таянии полярных ледяных шапок является спорным. В то время как исследования НАСА показали, что количество льда в Антарктиде на самом деле увеличивается, это увеличение составляет лишь треть от того, что теряется в Арктике.
Существуют убедительные доказательства того, что уровень моря повышается, а таяние арктических ледяных шапок является основной причиной. Со временем это может привести к обширным наводнениям, загрязнению питьевой воды и серьезным изменениям в экосистемах.
3. Transportation — Транспорт
Постоянно растущее население нуждается в транспорте, большая часть которого подпитывается природными ресурсами, выделяющими парниковые газы, такими как нефть. В 2014 году на долю транспорта пришлось 26% всех выбросов парниковых газов.
Транспорт также затрагивает другие экологические проблемы, такие как разрушение естественной среды обитания и увеличение загрязнения воздуха.
4. Natural Resource Use — Использование природных ресурсов
Недавние исследования показали, что человечество использует так много природных ресурсов, что нам потребуется почти 1,5 Земли для удовлетворения наших потребностей. Это будет только увеличиваться, поскольку индустриализация продолжается в таких странах, как Китай и Индия.
Увеличение использования ресурсов связано с рядом других экологических проблем, таких как загрязнение воздуха и рост населения. Со временем истощение этих ресурсов приведет к энергетическому кризису, а химические вещества, выделяемые многими природными ресурсами, вносят значительный вклад в изменение климата.
5. The Nitrogen Cycle — Круговорот азота
Поскольку основное внимание уделяется углеродному циклу, последствия использования человеком азота часто оказываются вне поля зрения. Предполагается, что сельское хозяйство может отвечать за половину связывания азота на земле, главным образом за счет использования и производства искусственных удобрений.
Избыточное содержание азота в воде может вызвать проблемы в морских экосистемах, в первую очередь из-за чрезмерной стимуляции роста растений и водорослей. Это может привести к заблокированным водозаборам и меньшему количеству света, попадающему в более глубокие воды, нанося ущерб остальному морскому населению.
6. Lowered Biodiversity — Снижение биологических ресурсов
Продолжающаяся человеческая деятельность привела к снижению биоразнообразия. Отсутствие биоразнообразия означает, что будущие поколения будут вынуждены бороться с растущей уязвимостью растений к вредителям и меньшим количеством источников пресной воды.
Некоторые исследования показали, что сокращение биологического разнообразия оказывает такое же сильное влияние, как изменение климата и загрязнение экосистем, особенно в районах с более высоким уровнем исчезновения видов.
7. Air Pollution — Загрязнение воздуха
Загрязнение воздуха становится все более опасной проблемой, особенно в густонаселенных городах. Всемирная организация здравоохранения (ВОЗ) установила, что 80% людей, живущих в городских районах, подвергаются воздействию уровня качества воздуха, который организация считает непригодным.
Это также напрямую связано с другими экологическими проблемами, такими как кислотные дожди и эвтрофикация. Животные и люди также подвержены риску развития ряда проблем со здоровьем из-за загрязнения воздуха.
8. Ocean Acidification — Закисление океана
Закисление океана — это термин, используемый для описания продолжающегося снижения уровня PH в океанах Земли в результате выбросов углекислого газа. Предполагается, что к 2100 году кислотность океана увеличится на 150%, если не будут предприняты попытки остановить ее.
Это увеличение подкисления может оказать страшное влияние на кальцифицирующие виды, такие как моллюски. Это вызывает проблемы во всей пищевой цепи и может привести к сокращению водной жизни, которое в противном случае не было бы затронуто подкислением.
9. Ozone Layer Depletion — Истощение озонового слоя
Истощение озонового слоя вызвано выбросом химических веществ, прежде всего хлора и брома, в атмосферу. Один атом может уничтожить тысячи молекул озона, прежде чем покинуть стратосферу.
Истощение озонового слоя приводит к увеличению УФ-излучения, достигающего поверхности Земли. Ультрафиолетовое излучение вызывает рак кожи, заболевания глаз, а также влияет на жизнь растений и на сокращение количества планктона в морской среде.
10. Acid Rain — Кислотный дождь
Кислотные дожди возникают в результате загрязнения воздуха, в основном из-за химических веществ, выделяющихся в окружающую среду при сжигании топлива. Его последствия наиболее отчетливо видны в водных экосистемах, где повышение кислотности в воде может привести к гибели животных.
Это также вызывает различные проблемы для деревьев. Хотя это не убивает деревья напрямую, кислотные дожди ослабляют их, повреждая листья, отравляя деревья и ограничивая доступное количество питательных веществ.
11. Overfishing — Чрезмерный рыбный промысел
По оценкам, 63% мировых рыбных запасов в настоящее время считаются истощенными. Это привело к тому, что многие рыболовные флоты направляются в новые воды, что приведет только к дальнейшему истощению рыбных запасов.
Чрезмерный вылов рыбы приводит к дисбалансу жизни океана, серьезно влияя на природные экосистемы. Кроме того, это также оказывает негативное влияние на прибрежные сообщества, которые полагаются на рыболовство для поддержки своей экономики.
12. Urban Sprawl — Застройка городов
Продолжающаяся экспансия городских территорий в традиционно сельские районы не обходится без проблем. Разрастание городов было связано с экологическими проблемами, такими как увеличение загрязнения воздуха и воды, в дополнение к созданию тепловых островов.
Спутниковые снимки, сделанные НАСА, также показали, как разрастание городов способствует фрагментации лесов, что часто приводит к увеличению обезлесения.
13. Deforestation — Вырубка лесов
Требования растущего населения привели к увеличению уровня обезлесения. Согласно текущим оценкам, планета теряет 80 000 акров тропических лесов в день.
Это приводит к потере среды обитания для многих видов, подвергая риску многие виды и приводя к крупномасштабному вымиранию. Кроме того, по оценкам, вырубка лесов дает 15% мировых выбросов парниковых газов.
14. Water Pollution Загрязнение воды
Пресная вода имеет решающее значение для жизни на Земле, но все больше источников загрязняются в результате деятельности человека каждый год. В глобальном масштабе ежедневно в воду попадает 2 миллиона тонн сточных вод, сельскохозяйственных и промышленных отходов.
Загрязнение воды может иметь вредные последствия вне загрязнения воды, которую мы пьем. Это также нарушает морскую флору и фауну, иногда изменяя репродуктивные циклы и повышая уровень смертности.
15. Population Growth — Рост населения
Многие из перечисленных здесь проблем являются результатом массового роста населения, который произошел на Земле в прошлом веке. Население планеты увеличивается на 1,13% в год, что составляет 80 миллионов человек.
Это приводит к ряду проблем, таких как нехватка пресной воды, потеря среды обитания для диких животных, чрезмерное использование природных ресурсов и даже исчезновение видов. Последнее особенно вредно, поскольку планета в настоящее время теряет 30 000 видов в год.
16. Waste Production — Производство отходов
Среднестатистический человек производит 4,3 фунта отходов в день, причем только на США приходится 220 миллионов тонн в год. Большая часть этих отходов попадает на свалки, которые производят огромное количество метана.
Это не только создает опасность взрыва, но метан также считается одним из худших парниковых газов из-за его высокого потенциала глобального потепления.
17. Genetic Modification of Crops — Генетическая модификация культур
Экологические проблемы, вызванные искусственными химикатами, становятся более ясными. Например, в Соединенных Штатах популяция бабочек-монархов сократилась на 90%, что может быть связано с убийцами сорняков, которые содержат глифосат.
Существует также предположение, что генетически модифицированные растения могут проникать через почву химических соединений в почву, что может повлиять на сообщества микроорганизмов.
The impact that human activities have on the environment around us is indisputable and more studies are being conducted each year to show the extent of the issue.
Влияние человеческой деятельности на окружающую среду неоспоримо, и каждый год проводится все больше исследований, чтобы показать как разрастаются экологические проблемы.
Читайте также:
- Нужна ли человеку мечта сочинение 10 предложений
- Сочинение на тему подольские курсанты
- Владимирские проселки солоухин сочинение
- Сочинение на тему помещичья русь
- Дельфин и человек сочинение
Our life is full of the modern technologies, that unfortunately cause serious damage to our nature. What can we do to salve this problem? The governments have been trying to reduce the pollution, but everyone should follow the next rules.
We should:
1. Walk more and use bicycle.
2. Use solar power to heat our homes.
3. Turn off lights, computers, TV and other electrical equipment when we’re not using them.
4. Encourage our parents to buy equipment that uses less electricity, including lights, air conditioners, heaters, etc.
5. Encourage our parents to buy electrical cars or the vehicles with low emissions.
6. Tell to our classmates, parents and teachers about what we know about this problem.
Let’s do these easy rules to help our environment!
Наша жизнь наполнена современными технологиями, которые, к сожалению, наносят серьезный ущерб нашей природе. Что мы можем сделать, чтобы разрешить эту проблему? Правительство пытается уменьшить степень загрязнения, но каждый должен знать следующие правила.
Мы должны:
1. Больше гулять и использовать велосипед.
2. Использовать солнечную энергию в качестве отопления наших домов.
3. Выключать свет, компьютеры, телевизоры и другое электрооборудование, когда мы их не используем.
4. Призывать наших родителей покупать оборудования, которые потребляют меньше энергии, включая лампочки, кондиционеры, обогреватели и др.
5. Призывать наших родителей покупать электромобили или автомобили с низким уровнем выхлопов.
6.Рассказывать нашим одноклассникам, родителям и учителям о том, что мы знаем об этой проблеме.
Давайте будем соблюдать эти простые правила, чтобы помочь нашей окружающей среде!
Ответственность за разрешение любых спорных моментов, касающихся самих материалов и их содержания,
берут на себя пользователи, разместившие материал на сайте. Однако администрация сайта
готова оказать всяческую поддержку в решении любых вопросов, связанных с работой и содержанием
сайта. Если Вы заметили, что на данном сайте незаконно используются материалы, сообщите об этом
администрации сайта через форму обратной связи.
Все материалы, размещенные на сайте, созданы авторами сайта либо размещены пользователями сайта
и представлены на сайте исключительно для ознакомления. Авторские права на материалы
принадлежат их законным авторам. Частичное или полное копирование материалов сайта без
письменного разрешения администрации сайта запрещено! Мнение администрации может не совпадать с точкой
зрения авторов.
- Главная
- Разное
- Образование
- Спорт
- Естествознание
- Природоведение
- Религиоведение
- Французский язык
- Черчение
- Английский язык
- Астрономия
- Алгебра
- Биология
- География
- Геометрия
- Детские презентации
- Информатика
- История
- Литература
- Математика
- Музыка
- МХК
- Немецкий язык
- ОБЖ
- Обществознание
- Окружающий мир
- Педагогика
- Русский язык
- Технология
- Физика
- Философия
- Химия
- Шаблоны, фоны, картинки для презентаций
- Экология
- Экономика
Содержание
-
1.
What we can do to solve the problem of acid rain? -
2.
2. These cars are working by water, solar power or electric battaries. -
3.
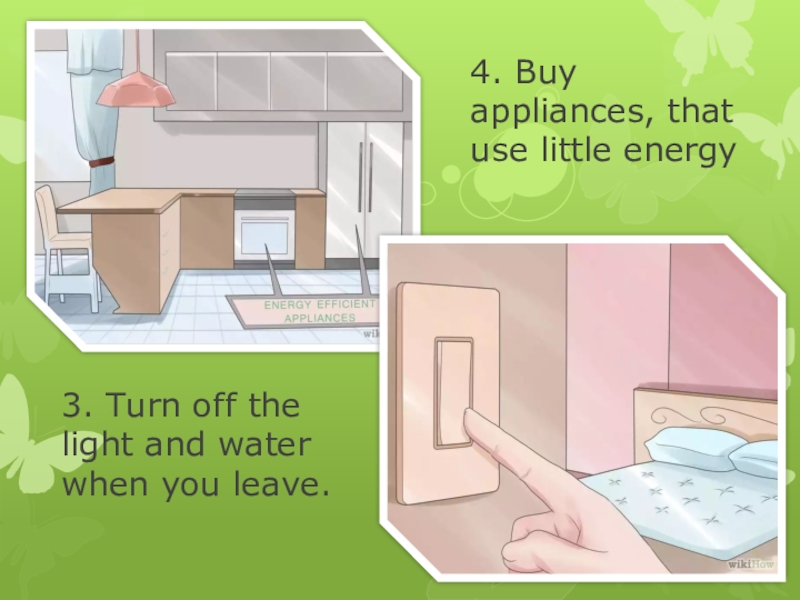
4. Buy appliances, that use little energy3. Turn off the light and water when you leave. -
4.
5. Try to buy less products of
2. These cars are working by water, solar power or electric battaries.
Слайд 1What we can do to solve the problem of acid rain?
Kuzmina
Katya 7A form

Слайд 22. These cars are working by water, solar power or electric

Слайд 34. Buy appliances, that use little energy
3. Turn off the light
and water when you leave.
Слайд 45. Try to buy less products of mass production, reducing the
use of factory materials.
6. Plant trees or your garden.
And keep animals!

The elevated levels of ions of hydrogen that land on the ground along with rainwater are what we all refer to as acid rain. There are many reasons for this phenomenon which have been studied by researchers over a long period of time. The acid rain is considered harmful not just for plants but for human life and other living beings as well. Hence it is all the more important for the students to read about the causes of acid rain, what harmful effects it has on us and other flora and fauna and finally what can be done to prevent it. Hence, we have come up with long essays for students on this very sensitive ecological topic, acid rain which would benefit students at large.
Contents
- List of Essays on Acid Rain in English
- Essay on Acid Rain – Essay 1 (150 Words)
- Essay on Acid Rain: Introduction, After-Effects and Conclusion – Essay 2 (250 Words)
- Essay on Acid Rain: Causes and Effects – Essay 3 (300 Words)
- Essay on Acid Rain – Essay 4 (400 Words)
- Essay on Acid Rain: Causes, Effects and Prevention – Essay 5 (500 Words)
- Essay on Acid Rain: With Conclusion – Essay 6 (600 Words)
- Essay on Acid Rain: Types, Sources and Harmful Impacts – Essay 7 (750 Words)
- Essay on Acid Rain: With Solution – Essay 8 (1000 Words)
List of Essays on Acid Rain in English
Essay on Acid Rain – Essay 1 (150 Words)
Introduction:
Acid rain simply means rain high in acidic contents such as a high rate of hydrogen ions. Acid rain is a cause of concern because of its effect on forest life and lakes. To a larger extent, acid rain is a big threat to our natural environment.
Causes of Acid Rain:
When we burn fossil fuel, we release a lot of sulfur dioxide and nitrogen oxide into the atmosphere. These compounds when mixed with air go straight into the atmosphere. At this level, they mix with water, oxygen, and other components to become acid rain.
Effect of Acid Rain:
For the aquatic animal, acid rain becomes deadly since it reduces the PH level in the water. Many animals that live in water need a particular PH level to survive. When that PH level is reduced, it becomes difficult to survive. For trees, acid rain leaves them exposed to diseases.
Conclusion:
Our animal and plant life should be of paramount importance to us. To end this, we must reduce our release of toxic gas into the atmosphere.
Essay on Acid Rain: Introduction, After-Effects and Conclusion – Essay 2 (250 Words)
Introduction:
Advancement of science and mass industrialization has led to many man-induced phenomenon and acid rain is one of them. It is nothing but rain that is highly acidic in content. Poisonous gases emitted by chemical industries pollute the atmosphere with oxides of sulphur, nitrogen and hydrogen. When these oxides raise high into the clouds, they react with water molecules there and turn them into sulphuric acid, nitric acid and hydrochloric acid respectively. These acids are released into the atmosphere as rain, snow or precipitation. Natural phenomenon like volcanoes and fossil fuel combustion also produce harmful gases which can cause acid rain.
After-Effects of Acid Rain:
Acid rains have a multitude of effects on humans, animals and the environment. Breathing problems like asthma is caused in children and adults alike. Skin peeling is possible when there is direct contact with acid rain. Corrosion of metals, peeling of paints on buildings, erosion of natural stones like marbles, granites and limestones are all inclusive. In addition, rivers and oceans get polluted with high contents of acid resulting in death of plants, fishes and other sea creatures. Fresh water reserves turn toxic and harmful to consume. Trees die due to direct and continuous exposure. Many insects are killed instantaneously. And exposed agricultural lands will no longer be fit for cultivation.
Conclusion:
Measures should be taken to avoid air pollution by industries. Environmentally friendly methods need to be adopted by everyone. Both the public and government need to realize the seriousness of the issue and work together to solve the problem of acid rain.
Essay on Acid Rain: Causes and Effects – Essay 3 (300 Words)
“Acid Rain”, according to the Royal Society of Chemistry this term was coined by Scottish chemist Robert Angus Smith in 1852. “Acid Rain” is a result of acidification of rainwater due to mixing relatively large quantities of oxides of sulphur and nitrogen emissions in the atmosphere. Interaction of this acidic water with other constituents of the atmosphere increases the soil acidity. It leaches away nutrients in the soil and heavily degrades quality of air. “Acid Rain” is posing a prospective threat to our ecosystem and environment.
Causes:
Rain water is increasingly acidified by pollutants released from homes, factories, power stations and cars.
i. Man-made Causes:
Heavy industrialization and urbanization is the major cause of acid rain. The exhausts of industries and factories emit harmful gases like Sulphur Dioxide (SiO2) and Nitrogen Dioxide (NO2) in the atmosphere. These gases interact with the water precipitation in the atmosphere and the chemical reaction between these gases and water molecules form Sulphuric Acid and Nitric acid.
A Large number of industries are set up nearby the majestic Taj Mahal, which emit harmful pollutants in the air and cause acid rain. The “Acid rain” reacts with the marble of Taj Mahal, causing damage to this heritage structure. To protect Taj Mahal over 2000 industries situated near Taj Mahal had transferred. The situation still remains critical because of the nearby leather industries.
ii. Natural Causes:
Volcano eruption releases various harmful gases in the environment which results in “acid rain” in the nearby areas.
Effects:
“Acid rain” causes heavy damage to the ecosystem in the affected areas. It has the largest impact on lakes, streams, wetlands, and other aquatic environments. Acidic water is absorbed into the soil and water bodies which turns them into toxic compounds. The toxic pollutants pose a huge risk to the survival of aquatic creatures like crayfish, clams, fish, and other aquatic animals. Forests are also destroyed by “acid rain” and the ecological balance of the affected area is hampered to a huge level.
Conclusion:
The only way to mitigate “acid rain” is to impose censorship on heavy industries which cause damage to our ecosystem. All the citizens must adopt and practise eco-friendly lifestyle. We must encourage use of alternative energy sources which will prevent burning of fossil fuels which releases harmful gases.
Essay on Acid Rain – Essay 4 (400 Words)
Acid rain is any form of precipitation which has acidic components like sulfuric acid or nitric acid. It falls to ground from the atmosphere in either wet or dry form. The precipitation can be in many different forms – rain, snow, fog, hail or even dust. It has a pH less than 5.6. Normally, the clean rain water has a pH of 5.6 as there is carbonic acid present in the rain water. This is because of carbon dioxide dissolved in water. The term acid rain was coined by Robert Angus Smith in 1872. There are two types of acid rain depositions – wet deposition and dry deposition.
It is a form of air pollution that is very damaging to the environment. The reason behind acid rain is particles like sulphur dioxide and nitrogen oxides which are emitted from fuel combustion. These molecules move up the atmosphere with the help of air currents and then they react with water and oxygen to form sulphuric acids and nitric acids. These acids combine with water before falling to ground in any form of precipitation. There are other sources of sulphur oxides and nitric oxides like volcanoes, manufacturing industries and vehicles. Even the decaying vegetation and wild forest fires generate the gases that form acid rain.
There are many negative effects of acid rain. Acid rain can make water bodies like lakes and ponds poisonous. This makes these water bodies inhabitable for marine life and makes the drinking sources unfit for human consumption. The number of aquatic animals and plants reduces because water becomes more acidic. It also degrades the quality of soil because of the acidic pH when it is absorbed into soil. As the soil quality is affected, it leads to reduction in the crop yield. There is a reduced growth of plants and trees in areas that are affected by acid rain. It impacts human health by causing skin problems, heart and lung issues. Other than biotic components, it affects and damages buildings and property. It is corrosive in nature and this is reflected in the corrosion of Taj Mahal by acid rain.
The problem is increasing and becoming worse because of the rapid population growth and industrialization. Although natural sources cannot be contained or regulated, we can at least work on controlling the manmade causes. Acid rain can be reduced by choosing cleaner forms of energy like solar power. Afforestation is another thing that can help in reducing air pollution.
Essay on Acid Rain: Causes, Effects and Prevention – Essay 5 (500 Words)
Introduction:
Acid rain is basically wet deposition of acids in rain or snow that is of low pH on to the surface of the earth. However, rain water is naturally acidic due to the carbonic acid rain is an environmental hazard brought about by pollution. Pollution of the environment especially air and water pollution result in formation of acid rain.
Formation and Causes of Acid Rain:
Acid rain occurs in two ways. Wet deposition occurs when there is a precipitation and raindrops deliver the acidic components to the earth’s surface whereas dry deposition occurs when no precipitation occurs and the pollutants stick to plants and the earth surface. Air pollution causes the accumulation of chemicals in the atmosphere. These chemicals form chemical reactions that react with water and oxygen in the atmosphere and form acid rain. Gases that are oxides like Sulphur dioxide and nitrogen oxide form reactions with oxygen and water and form acids. These chemical reactions increase with the increase in air pollution by human or natural activities. Natural activities like the eruption of volcanoes or hot springs in geothermal result in the release of Sulphur dioxide to the atmosphere while lightning strikes cause a release of nitrogen oxide to the atmosphere. Human activities especially industrialization, combustion of fuels and emission of greenhouse gases to the atmosphere also contribute to the accumulation of oxides in the atmosphere which result in acid rain formation. Air pollutants have greatly increased the formation of acid rain and the effects have affected the health of living things on the surface of the earth.
Effects of Acid Rain:
Acid rain affects both animate and inanimate components of the environment. Plants and animals are mostly affected health wise because of the direct interaction. Plants get scorched and growth is impaired because acid rain also affects the soil pH by acidifying it. Animals, including humans consume acid rain when harvested which affects their health negatively especially through heart and lung problems. Acid rain is deleterious to aquatic animals like fish because acidic water flows to water bodies. Inanimate objects like buildings are also negatively affected. Buildings and bridges made of stone, steel or sand are corroded by the acids.
Prevention of Acid Rain:
Reduction of pollutants released to the atmosphere will prevent acid rain formation. The industries will have to ensure that filtration of gases before release to the atmosphere is done. There is a technical method of flue-gas desulphurization that helps in preventing emissions of Sulphur dioxide and compounds to the atmosphere. The adherence to air quality agreements that have been put in place is important. Government effort in gaseous emissions control programs will enable the regulation of pollutants released to the atmosphere therefore acid rain formation will be reduced.
Conclusion:
In conclusion, acid rain is an environmental hazard that is caused by acidic components that accumulate in the atmosphere. Acid rain has both deleterious and adverse effects to the environment. The control of air pollution will result in reduction of acid rain formation.
Essay on Acid Rain: With Conclusion – Essay 6 (600 Words)
Introduction:
‘Acid Rain’ is not a term that we hear very commonly. Most of us would not even know that such a type of rain exists. Before we move any further, let’s first understand what acid rain means.
The literal term ‘Acid Rain’ will make us think that acid would pour from the sky as rain but it is not so. Acid rain is formed when certain gases mix up with the atmospheric moisture and create precipitation which is more acidic than the normal one. In chemical terms, when the pH level of rain water falls below 5.6, it turns acidic and changes as acid rain. Robert A. Smith was the first to use the term ‘Acid Rain’ in 1872 in his studies of air in Manchester, England. But the term was most widely recognized only in 1980.
Effects of Acid Rain:
On a general note, rain is welcomed by all since it refreshes the atmosphere and benefits the environment. But not all rains do this and acid rain is a best example to show the devastating effects that a rain can cause. Since most of us are unaware of the subject acid rain, we have only little knowledge about it. Acid rain can destroy anything over a short period of time. Acid rain is very dangerous to the environment.
Acid rain falling on rivers, lakes and other water bodies makes it poisonous and destroys marine life.
It damages the forests and kills the insects and other living creatures. Black Forest in Germany received its name due to the acid rain which turned the trunks and branches of all its trees into black.
Acid rain causes severe damage to buildings and monuments because these are made of Calcium Carbonate stone; acid rain reacts with Calcium Carbonate and destroys it. The paint peels off quickly and the statues lose their good appeal. Taj Mahal, one of the wonders of the world has faced the ill-effects of acid rain.
When the acid rain falls on a place, it changes the acidity level of that place causing great harm to both the living and non-living things existing there.
Acid rain is hazardous to humans also as it causes respiratory problems and various other health issues.
Causes of Acid Rain:
Both nature and humans are to be blamed for the occurrence of acid rain.
Normal rain occurs when Carbon dioxide and water present in the air react together and forms Carbonic acid which is a weak acid. Acid rain is caused when Sulphur dioxide and Nitrogen Oxide reacts with water molecules in the atmosphere and produces acid which is harmful in nature. So, the main cause of acid rain is the emission of Sulphur dioxide and Nitrogen oxides in the atmosphere. This emission occurs in two ways – emission through volcanic eruption and decaying vegetation, combustion of fossil fuels from transport, burning chimneys and industrialization.
Ways to Prevent Acid Rain:
Acid rain is very dangerous and it should be stopped from occurring. We cannot stop acid rain that occurs due to natural reasons but we can surely do our best to reduce the use of fossil fuels which leads to the emission of gases like Sulphur dioxide and Nitrogen Oxide. Air pollution is the main reason for almost all the environmental problems and humans should make all attempts to reduce pollution. Using solar power instead of electricity, recycling natural resources and planting more trees can help in purifying the air and can also help in preventing man-made disasters like acid rain.
Conclusion:
Each of us must take vow to preserve the natural resources and maintain the environment as clean and healthy as possible. Causing damage to the environment will only lead to the extinction of this Planet and no technology can help us buy a beautiful home similar to Earth.
Essay on Acid Rain: Types, Sources and Harmful Impacts – Essay 7 (750 Words)
Introduction:
Acid Rain simply means a rainfall that is highly acidic and such rain causes environmental, atmospheric and material depletion. Acid rain affects infrastructure, aquatic creature, plants and so many more. When something is acidic, it means it has a high level of ions of hydrogen which also means that the pH of the object or substance is low. Rain water that is normal is already a little bit acidic and has pH between 5.3 and 6.0. This is so because the water and carbon dioxide that are together in the atmosphere react together and form an acid known as carbonic acid; carbonic acid is an acid that is weak. Whenever the pH of water drops below the above stipulated range, the rain water is said to be acid rain.
Acid rain formation isn’t due to just one reason; both man-made and natural sources are believed to influence acid rain. That said, the emissions and release of nitrogen oxides and sulphur dioxide as a result of fossil fuels being combusted is the major cause of acid rain. Acid rain can be in form of snow, rain, fog, hail or even dew that contains high level of acid pollutants, most especially nitric acid and sulphuric acid. When emitted nitrogen oxide and sulphur dioxide reacts and mix with water molecules that are present in the air and atmosphere, acid rains are produced.
Evolution of Acid Rain:
The name “acid rain” was given by a man called Robert Smith in 1872. In recent years, industrialisation and population has led to a sharp and exponential increase in acid rains with it becoming a lot more alarming. Acid rains are also being further influenced by the use of chimneys that are very tall also known as smokestacks on ships, factories, etc. Smokestacks are used to lower the rate of air pollution but they end up spreading and causing acid rains through the release of gases right into the atmospheric circulation of a region. There is a very large quantity of deposits of acid in the United States, Canada and almost all of Europe including parts of Germany, Norway and Sweden. Of recent, some parts of the southern part of Asia, Sri Lanka and South Africa have also witnessed deposits of acid.
Types of Acid Rain:
Acid rains are divided based on the type of deposition by which the occurrence of the acid rain takes place.
Basically, we have two depositions types and they are discussed below:
Wet Deposition:
When acid rain is in form of snow, rain, mist or fog when it falls to the surface of the earth, acid is removed from the air or atmosphere by these liquids and is dropped on the surface of the earth. The flow of this acid rain through the earth affects a great quantity of aquatic life, animals and plants. Water moves from the drains into small water bodies like canals and rivers which in turn flow into seas and ocean causing damage to marine life.
Dry Deposition:
When there is a merging together of various acidic pollutants and they form smoke and dust that comes to the surface of the earth in the form of dry particles that stick on the surface of the earth and many other surfaces like monuments, trees, houses, cars and buildings. It is important to note that almost all of pollutants that are in the air or atmosphere are spread through the method of dry deposition.
Sources of Acid Rain:
Acid rains are majorly caused by two sources and the sources are discussed below:
1. Natural Sources:
Volcanic eruptions are the major agents that cause acid rain. Huge quantities of lava that produces gases that are harmful are emitted by volcanoes and this causes acid rain that are quite more severe than usual. The gases that form the acid rain are also generated by biological processes including wildfires and decaying vegetation that occur within the environment.
2. Human Sources:
Activities of humans that lead to the release of gases like nitrogen and sulphur from automobile plants, power generating setups and factories are the causes of acid rain here. Coal when it is used to generate electric power is probably the greatest contribution to the emission of gases that can cause acid rain
The harmful impacts of Acid Rain:
Acid rain can bring serious harm to the environment and a few of the various wide segments of the environment where it can wreak havoc are listed below:
1. Public health
2. Wildlife and forests
3. Infrastructure and architecture
4. Soil
5. Marine life.
Essay on Acid Rain: With Solution – Essay 8 (1000 Words)
The presence of harmful hydrogen ions in the raindrops which fall on us is called acid rain. Acid rain is a prominent term alluding to the testimony of a mixture of wet (rain, snow, slush, mist, cloud water, and dew) and dry (acidifying particles and gases) acidic segments. Fluids with a pH under 7 are termed acidic, and those with a pH more prominent than 7 are alkaline.
History of Acid Rain:
The destructive effect of polluted, acidic nature of the air of a region on limestone and marble was noted in the seventeenth century by John Evelyn, who commented upon the poor state of the Arundel marbles. Since the Industrial Revolution, outflows of sulphur dioxide and nitrogen oxides into the environment have increased. In 1852, Robert Angus Smith was the first to demonstrate the connection between acid rain and climatic contamination in Manchester, England. He is also called as the Father of Acid rain due to his immense contribution towards the study of acid rain. Despite the fact that acid rain was found in 1853, it was not until the late 1960s that researchers started generally watching and observing the phenomenon. The expression acid rain was coined in 1872 by Robert Angus Smith.
Causes of Acid Rain:
The cause of acid rain is attributed to both man-made as well as natural causes.
Natural Causes – The major natural cause leading to acid rain is the volcanic eruptions. Volcanoes emanate acid releasing gases to make higher than ordinary measures of acid rain or some other type of precipitation, for example, mist and snow to a degree of influencing vegetation cover and wellbeing of inhabitants in an area. Decaying vegetation, out of control fires and natural procedures inside the earth likewise produce the acid rain-causing gases. Lightning strikes in addition normally create nitric oxides that respond with water particles by means of electrical action to deliver nitric acid and thus causing acid rain.
Man-Made Causes – Human activities prompting substance gas outflows, for example, sulphur and nitrogen are the essential causes of acid rain. The activities incorporate air contamination sources transmitting sulphur and nitrogen gases like power generation facilities and vehicular emissions. Specifically, utilization of coal for electrical power is the greatest cause of vaporous emanations prompting acid rain. Vehicles and manufacturing plants likewise discharge high scores of vaporous emanations on consistent schedule into the air, particularly in exceedingly industrialized zones and urban areas with huge quantities of vehicle movement. These gases respond in the air with water, oxygen, and different synthetic concoctions to shape different acidic mixes, for example, sulphuric acid, ammonium nitrate, and nitric corrosive. Accordingly, these areas encounter exceedingly high measures of acid rain.
Effects of Acid Rain:
Acid rain affects almost everything. Plants, soil, trees, structures and even statues can be changed by the precipitation. Acid rain has been observed to be very harmful to the trees. It debilitates them by washing ceaselessly the defensive film on leaves, and it stunts development. Acid rain can likewise change the creation of soil and waterways, making them appalling for nearby creatures and plants. For instance, normal lakes have a pH of 6.5 or higher. As acid rain raises the dimension of corrosiveness, the waters turn acidic. Most fish species can’t endure a water pH of underneath 5. At the point when the pH turns into a 4, the lake is viewed as dead. Such is the effect an acid rain can have on life. It can furthermore disintegrate and corrode limestone and marble structures and landmarks, similar to tombstones.
Solutions Ahead:
There are a few answers for ceasing artificial acid rain. Controlling the emanations originating from vehicles and structures is an essential step. This should be possible by confining the utilization of petroleum derivatives and concentrating on progressively maintainable vitality sources, for example, solar energy and wind control.
Additionally, every individual can do their part by decreasing their vehicle use. Utilizing open transportation, strolling, riding a bicycle or carpooling is a small yet important step. Individuals can likewise decrease their utilization of power, which is broadly made with petroleum by-products, or change to a sunlight based arrangement. Numerous power organizations offer sun oriented packs to their clients that require no establishment and low expenses.
Acid Rain Instances in India:
It has been discovered that potential neutralizer of the acidic parts of rainwater in the Indian subcontinent is Calcium which is predominantly available from the soil. As the soil of the most part of Indian land is Calcareous, it contains a bounty of calcium. Taj Mahal, one of the most beautiful historical monuments has been affected by acid rain. The city of Agra has numerous enterprises which transmit the oxides of sulphur and nitrogen in the environment. Individuals keep on utilizing low-quality coal and kindling as household fuel, adding to this issue. There have hardly been any measures taken in order to have a check on this increasing pollution and industrialization in Agra. Acid rain caused in this region is due to the interaction of sulphuric acid with calcium carbonate resulting in the formation of calcium sulphate. This has led to the damage of the surface of Taj Mahal and it has lost its shine owing to this acid rain.
Conclusion:
There are natural causes of acid rain which are beyond our control. However, there are enough causes which have been instigated by us and we have all the means in this world to control them. What is, however, required is our willingness to do so. Only if we decide today that we are going to stop or at least minimise the use of petroleum by-products and instead switch to cleaner fuels, we can prevent a lot of current environmental issues such as air pollution, water pollution and acid rain. It is for all of us to understand that we need to this not for plants and animals but for our own good and for the betterment of our future generations. Some small measures by all individuals along with some measures in place by the government can do wonders for our country. At least we can make it a better place to live for all.
Message from Sophia!
I would love to hear from you and read your comments on this article. Let me know what you think about this article. Is it helpful to you? Your comments and suggestions will serve as an inspiration and learning platform for me.
Regards, Sophia
Although the cause and effect relationship between emissions of SO2 and NOX and acid rain damage is not universally accepted, most of the solutions proposed for the problem involve the disruption of that relationship. The basic approach is deceptively simple. In theory, a reduction in the emission rate of acid forming gases is all that is required to slow down and eventually stop the damage being caused by the acidification of the environment. Translating that concept into reality has proved difficult, however.
The reduction in emissions of SO2 and NOX is a long-term solution, based on prevention. It is also a solution in which the environment itself has a major role. As emissions decline, it must adjust until some new level of equilibrium, reflecting the decreased acidity, is attained. There is concern that in some areas the damage has gone too far to be reversed completely, and there is currently some support for this point of view. For example, SO2 emissions in Britain have declined by nearly 40 per cent since 1970 (Caulfield and Pearce 1984), and, in Canada, emissions decreased by 45 per cent between 1970 and 1985 (Environment Canada 1991), yet the reduction in aquatic or terrestrial acidity downwind from these areas remains less than expected. The discrepancy may be explained in part by the continuing rise in NOX emissions, offsetting the decline in SO2, but it is also possible that the link between reduced acidity and ecological recovery is not linear. In that case, a specific reduction in acid emissions might not bring about an equivalent reduction in environmental damage (Park 1991). In Lake Oxsjon in Sweden, for example, the pH fell from 6.8 to 4.5 between 1968 and 1977. Despite the marked reduction in SO2 emissions from Britain and Sweden in the decade or so since then, the pH has only recovered to 4.9 (Mason 1990). Reductions of a similar magnitude have been identified in some Scottish lochs (Last 1989). Surveys in lakes in Ontario indicate that pH values may rise by small but significant amounts soon after acid input is reduced, but it may be 10 to 15 years before aquatic biota respond to the reduced acidity (Havas 1990). This indicates that natural recovery does seem to be possible, but that it is a slow process, taking much longer than the original acidification. It may also indicate that some direct human input will be necessary, either to initiate the recovery process or to speed it up.
One possible input is the addition of lime, which would produce an immediate reduction in acidity, and allow the recovery mechanisms to work more effectively. Lime has been used as a means of sweetening acid soils for many years, and may be the reason that in areas of acid soils agricultural land is less affected by acid rain than the natural environment. In areas where natural regeneration is no longer possible, the restoration of the original chemical balance of the soil by liming and appropriate fertilizer application might allow reforestation to be successful.
The same situation might apply in the aquatic environment. Simply re-stocking an acidified
Figure 4.12 Reduction of sulphur dioxide through emission controls
Figure 4.12 Reduction of sulphur dioxide through emission controls
- Source: Various; see text
lake with fish cannot be successful unless some buffering agent is added. In 1973, several lakes in the Sudbury area were treated with calcium carbonate and calcium hydroxide in an attempt to reduce acid levels (Scheider et al. 1975). Acidity returned to normal and there was an increase in nutrient levels, but, in the lakes closest to Sudbury, copper and nickel remained at concentrations toxic to fish (Ontario: Ministry of the Environment 1980). Similar experiments in Sweden since the mid-1970s have involved the liming of some 3,000 lakes and 100 streams, and have provided encouraging results (Porcella et al. 1990). Reduced acidity is often followed by recovery among the aquatic biota, Lower organisms—such as phytoplankton—which reproduce rapidly recover first, followed eventually by amphibians and fish (Havas 1990). Artificial buffering of lakes in this way is only a temporary measure which may be likened to the use of antacid to reduce acid indigestion. The neutralizing effects of the lime may last longer than those of the antacid, but they do wear off in 3 to 5 years and re-liming is necessary as long as acid loading continues (Ontario: Ministry of the Environment 1980). The treatment of the environment with lime to combat acidity is only a temporary measure, at best. It can be used to initiate recovery, or to control the problem until abatement procedures take effect, but since it deals only with the consequences of acid rain rather than the causes, it can never provide a solution.
Most of the current proposals for dealing with acid rain tackle the problem at its source. They attempt to prevent, or at least reduce, emissions of acid gases into the atmosphere. The only way to stop acid emissions completely is to stop the smelting of metallic ores and the burning of fossil fuels. Modern society could not function without metals, but advocates of alternative energy sources—such as the sun, wind, falling water and the sea—have long supported a reduction in the use of fossil fuels. These alternative sources can be important locally, but it is unlikely that they will ever have the capacity to replace conventional systems. Nuclear power has also been touted as a replacement, since it can be used to produce electricity without adding gases to the atmosphere. It has problems of its own, however. Difficulties associated with the disposal of radioactive wastes remain to be resolved, and events such as the Chernobyl disaster of 1986 do little to inspire public confidence in nuclear power. Thus, although the replacement of fossilfuel-based energy systems with non-polluting alternatives has the potential to reduce acid rain, it is unlikely to have much effect in the near future.
Since SO2 makes the greatest contribution to acid rain in North America and Europe, it has received most attention in the development of abatement procedures, whereas emissions of NOX—which are both lower in volume and more difficult to deal with—have been largely neglected. Similarly the development of control technology has tended to concentrate on systems suitable for conventional power stations, since they are the main sources of acid gases (Kyte 1986a). Sulphur dioxide is formed when coal and oil are burned to release energy, and the technology to control it may be applied before, during or after combustion. The exact timing will depend upon such factors as the amount of acid reduction required, the type and age of the system and the cost-effectiveness of the particular process (see Figure 4.12).
One of the simplest approaches to the problem is fuel switching, which involves the replacement of high sulphur fuels with low sulphur alternatives. This may mean the use of oil or natural gas rather than coal. In Britain, for example, the recently privatized power industry is actively exploring the increased use of North Sea gas as a means of reducing SO2 output, despite concern that this approach is a waste of a high premium fuel with a relatively short lifespan (Stevenson 1993). However, since most power stations use coal and are not easily converted to handle other fuels, fuel switching usually involves the replacement of one type of coal with another or even the blending of low and high sulphur coal. Much depends on the availability of the low sulphur product. In Britain, for example, the supply is limited (Park 1987), but in western Canada and the western United States, abundant supplies of low sulphur coal are available with a sulphur content only one-fifth of that which is normal in eastern coal (Cortese 1986). Such a difference suggests that fuel switching has a considerable potential for reducing SO2 production, yet wholesale substitution is uncommon. The problem is a geographical one. The main reserves of low-sulphur coal are in the west, far removed from the large consumers in the east. Transport costs are therefore high, and complete switching becomes economically less attractive than other methods of reducing SO2 output. Compromise is possible. Rather than switching entirely, Ontario Hydro, the major public electricity producer in the province, has a well-established practice of blending low-sulphur, western Canadian coal, with the high-sulphur product from the eastern United States. As a result of this plus the use of washed coal, the utility’s SO2 output per unit of electricity has been declining with some regularity since the early 1970s (Ontario: Ministry of the Environment 1980).
The amount of SO2 released during combustion can be reduced if the coal or oil is treated beforehand to remove some of the included sulphur, in a process called fuel desulphurization. The methods can be quite simple and quite cost effective. Crushing and washing the coal, for example, can reduce subsequent SO2 emissions by 8 to 15 per cent (Park 1987), which represents a reduction of 1.5 to 2 million tonnes of SO2 per year in the eastern United States alone (Cortese 1986). More complex chemical cleaning methods involving the gasification or liquefaction of the coal are also possible, but at considerable cost (Ramage 1983).
If it is not possible to reduce sulphur levels significantly prior to combustion, there are techniques which allow reduction during the combustion process itself. Basically, they involve the burning of coal in the presence of lime. Although the technology has been studied since the 1950s, it has yet to be adopted on a large scale (Ramage 1983). There are two promising developments, however, which are expected to be available in the early 1990s. These are, lime injection multi-stage burning (LIMB) and fluidized bed combustion (FBC). LIMB involves the injection of fine lime into the combustion chamber, where it fixes the sulphur released from the burning coal to produce a sulphate-rich lime ash. This process can reduce SO2 emissions by 35-50 per cent (Burdett et al. 1985). In the FBC system, air under pressure is injected into a mixture of coal, limestone, and sand, until the whole mass begins to act like a boiling fluid (Ramage 1983). The continual mixing of the materials under such conditions ensures that combustion is very efficient, and that up to 90
per cent of the sulphur in the fuel is removed (Kyte 1986b). It has the added advantage that, since furnace temperatures are relatively low, it reduces NOX emissions also. In the United States, four thermal electric generating stations utilizing FBC technology came on stream in the late 1980s. Together they produce only 400 MW, but it has been estimated that a further 150 stations— producing 20,000 MW—could be retrofitted (Ellis et al. 1990).
Flue gas desulphurization (FGD) is the name given to a group of processes which remove SO from the gases given off during combustion. The2 devices involved are called scrubbers, and may be either dry or wet operations. The simplest dry scrubbers act much like filters, removing the gas on contact by chemical or physical means. Sulphur dioxide passing through a dry pulverized limestone filter, for example, will react chemically with the calcium carbonate to leave the sulphur behind in calcium sulphate (Williamson 1973). Other filters—such as activated charcoal—work by adsorbing the gas on to the filter (Turk and Turk 1988).
Wet scrubbers are more common than the dry variety. The flue gases may be bubbled through an alkaline liquid reagent, which neutralizes the SO2 and produces calcium sulphate in the process (Kyte 1981). A variation on this approach involves the use of a lime slurry through which the flue gases are passed, and in more modern systems the combustion gases are bombarded by jets of lime (LaBastille 1981) or pass through spray systems (Ramage 1983). The system described by LaBastille removes 92 per cent of the SO2 from the exhaust gases and many scrubbers achieve a reduction of between 80 and 95 per cent (Cortese 1986). By 1978, Japanese industry had installed scrubbers on more than 500 plants (Howard and Perley 1991). In the United States, some 70,000 MW of electricity is currently being produced from plants employing FGD technology, and in Canada, a FGD retrofit programme is in place with the goal of reducing SO2 emissions by about 50 per cent by 1994 (Ellis et al. 1990). The European Community directive of 1988—requiring all EC nations to reduce SO2
emissions by stages up to 70 per cent by 2003— will be met in large part by the installation of FGD equipment (Park 1991).
Flue gas desulphurization is thus one of the most common methods of SO2 removal, in part because of the high efficiencies possible, but for other reasons also. Scrubbers are technically quite simple, and can be added to existing power plants relatively easily. Retrofitting existing plants in this way is less expensive than building entirely new ones, and in some systems the recovery of sulphuric acid for sale can help to offset the cost.
None of the FGD systems described works well to reduce emissions of NOX from power plants. The best results for NOX control—up to 80 per cent reduction—have been obtained using a selective catalytic reduction process (SCR), which breaks the NOX down into the original N and O, but the price is high. To retrofit an existing thermal power station would cost an estimated $10,000 for every 1,000 kg of NOX removed, and maintenance costs would be substantial because the life of the catalyst is short (Ellis et al. 1990). Difficulties also exist with emission reductions from automobile exhausts. The development of technology that can produce a cooler burning internal combustion engine, or perhaps replace it completely, may be required before emissions of NOX from that source are reduced significantly (Park 1987).
It is now technically possible to reduce SO2 emissions to very low levels. However, as is common with many environmental problems, technology is only one of the elements involved. In many cases, economics and politics have retarded the implementation of technical solutions
Figure 4.13 Technologies and trade-offs in power station emission control
Source: From Ridley (1993)
Figure 4.13 Technologies and trade-offs in power station emission control
to environmental problems, and that is certainly the case with acid rain.
Continue reading here: The Economics And Politics Of Acid Rain
Was this article helpful?



















